4 - Orbitals, Shells and Subshells
Abstract (TL;DR):
This is an introduction to the first three of four quantum. The first, the principle quantum number, demonstrates the energy level of the atom represented by its electron shells. The second, the orbital quantum number, demonstrates the electron subshells, or the particular configurations electrons take in three dimensions. The third, the magnetic quantum number, determines how many subshells there are within an orbital.
—
Image by MooMooMath and Science
I know how much you were looking forward (or perhaps dreading) this part. Well, if not “looking forward to”, perhaps still trying to reason how all of this is going on within every element in your body and facing a slight existential crisis. All I have to say, in that case, is to try not to worry about it, and, of course, prepare for another dive into the boundary of quantum mechanics and chemistry.
I recommend that you check out the previous parts of the Elemental Flow series, especially Part 2. Familiarizing yourself with the quantum mechanics of electrons before this lesson will make this a smooth experience.
Quantum Numbers
What do the letters “s”, “p”, “d”, “f”, Aufbau, spin, and shapes have in common?
You might be scratching your head and wondering, “Why is he asking me this when I know he’s going to tell me.” And you’re right. Let’s break out our handy dandy…periodic table. And notebook, if you have one (I got you, 90s kids).
Image via Science Notes
Orbitals are organized according to their energy levels. Naturally, every element on the periodic table must exist at a different energy state in nature than each other (otherwise, what you learned in Part 1 and Part 2 would be meaningless – electrons have no need to configure themselves in different ways if every atom exists at the same energy level). Therefore, there must be a different orbital for each element. And, indeed, there is. The periodic table shows that, at the bottom of each element, there is a different electron configuration for each element. This exclusivity in configuration is called the Pauli Exclusion Principle.
Attentive readers might have already noticed the letters at the bottom of the elements on the periodic table – “s”, “p”, “d”, and “f”.
These labels are only descriptors of one of four quantum numbers, which are certain quantities within a quantum mechanical system. Simply put, since electrons are quantum mechanical systems, they hold specific quantities.
Electrons have four such quantities. We call them the principal quantum number, or n, the orbital quantum number, or l, the magnetic quantum number, or m, and the spin quantum number, or s.
The Quantum Dive – Breaking the Shells
1) Principal Quantum Number (n)
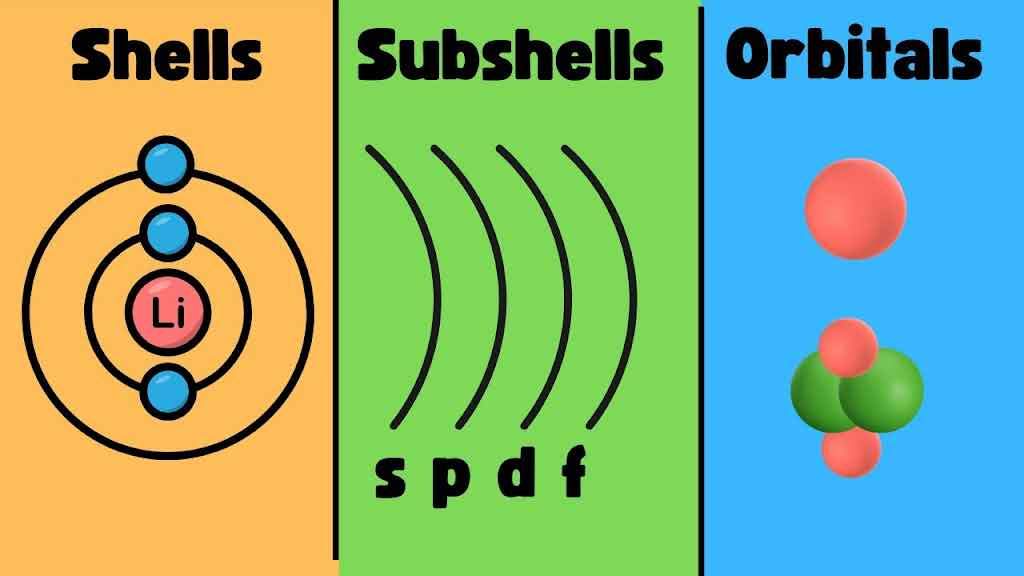
The first, n, is the number that labels the distance from the nucleus, or the energy level of an electron. Since we know, from Part 1, that the electrons with the lowest energy are the ones closest to the nucleus, we can tell that the lower the value of n, the lower the energy level. In chemistry, the energy level of an electron is described as its electron shell.
These shells are labeled from K to O starting from their innermost shell to their outermost shell, according to their principal quantum number. To be clear, an n of 1 is referred to as the K shell, n = 2 is L, n = 3 is M, n = 4 is N and n = 5 is O. While they do have this alphabetical label, when describing shells, more people recognize the numeric value of n. However, they can help you organize when we talk about the subsequent quantum numbers.
2) Orbital Quantum Number (l)
Next up, l. This quantum number explains the orbital angular momentum, or the momentum brought about by movement of electrons around their nucleus.
Before I move on from here, I did want to explain this number more carefully. To do so, we’ll need to review.
Electrons, to remain stable, exist as standing waves. These standing waves exist in three dimensions, meaning the peaks and troughs of this wave motion move not only toward and away from the nucleus, but also around the nucleus. At the lowest energy levels, brought about at n = 1, the electron motion is, therefore, spherical around the nucleus (which we see on the electron cloud model for hydrogen).
When more electrons are added, things get interesting. In these cases, electrons begin to influence each other, since they’re all negatively charged. However, they’re still all attracted to the positively charged atomic nucleus. What do they do? Well, like a game of Twister, they orient themselves into different shapes so that they can maintain their attraction to the nucleus, while minimizing the impact from and on other electrons. These “shapes” are driven by how electrons must move around the nucleus to achieve the lowest possible energy levels. These shapes are what are known as orbitals.
Now, you see where the name of this quantum number comes from. However, it's also known by another name: the azimuthal quantum number. This also provides another way of understanding this quantum number.
Within our three-dimensional world, we can describe the position of anything in relation to anything else. In astronomy, the azimuth and altitude give the direction of a star in this spherical system in relation to some observer at the center.
Electrons aren’t a single point, like a star. They have the wave motion that we explained earlier. As such, they move around the “wall” of the zenith in three dimensions (hence, the spherical shape of the first principle quantum number, n = 1).
The mathematical calculation behind finding an electron’s orbital angular momentum results in non-negative integers, like 0, 1, 2, 3, etc. (these are the orbital quantum numbers). An n of 1 and an l of 0 is represents the spherical orbital described previously, the lowest energy state of an atom, held by hydrogen. Thanks to Pauli’s exclusion principle, only hydrogen has this quantum state.
The orbital quantum number on the periodic table is represented by the letters s, p, d, and f (although, as more elements are created, there may be more letters added), where s is l = 0, p is l = 1, d is l = 2, and f is l = 3. Each number has a certain number of shapes, creating another grouping called electron subshells.
You’ll notice that there can be multiple subshells in one shell. Each individual electron configuration is called an orbital.
Image by thomji via Chemistry Stack Exchange
3) Magnetic Quantum Number (m)
Before we wrap up with this image, there’s something important in it that will help you understand the magnetic quantum number.
You might have noticed above that there are three different p orbitals in the above image. That is due to the different ways an orbital can orient itself within space as the amount of electrons in the system increases. This is what Bohr did not account for in his model; when more electrons enter the system, in order to keep energy as low as possible, the system must orient itself differently within the three dimensions.
The magnetic quantum number is a handy way of telling us how many ways a subshell can orient itself. I say it’s handy because there’s a simple equation to tell you how.
For every value of l, there is a range of values of m from -l to l, including zero.
So, if our system is a d orbital, which is l = 2, there would be 5 ways the subshells could orient themselves, given by the m values -2, -1, 0, 1 and 2.
That’s all there is to that. See? Not so bad.
Intermission
Okay, okay. Break time. We don’t want to overcook your brains with chemistry and quantum theory. Fortunately, what we are going over is, theoretically, all there is to orbitals (without the mathematics, of course). And, as you already know, this leads to some amazing science. After all, after learning about electron shells, the next step can only be how elements combine to reduce energy even further!
Are you enjoying learning about how these microscopic particles work and seeing how something so complex can create something as simple as a drop of water and/or something as ubiquitous as air?
Let me know. I would love to talk science with you guys.
As always, thank you for joining me, and I will see you again very soon.